
Infections of the middle ear – the air-filled space behind the eardrum that contains the tiny bones essential to hearing – are believed to affect more than 700 million people worldwide every year. Many require surgery, and the insertion of ear tubes is a common procedure. In the US, more than 600,000 children each year have tympanostomy tubes (TT) implanted. TTs are commonly used to treat otitis media, a middle ear disorder that frequently occurs in infants and children. It presents with earaches, impaired hearing, and fever, and if antibiotics or decongestants are ineffective, a myringotomy to create a slit in the tympanic membrane might be performed to aerate the middle ear and remove fluid. If the problem persists, insertion of a TT can create continuous aeration.
Children are especially prone to ear infections – Harvard Medical School reports that 40% develop recurrent or chronic infections – creating risk of impaired hearing, speech and language delays, perforation of the eardrums, or even meningitis.
The success of these operations is crucial, so one would think that any major problems with TTs would have been resolved through advances in technology, but this is not the case.
“The design of TTs is an important and popular conversation point and there is an impetus to innovate in this space,” says Dr Aaron Remenschneider, lecturer in head and neck surgery at Harvard Medical School and Investigator at Massachusetts Eye and Ear (MEE).
“Many children have persistent ear infections or fluid behind the eardrum, and TTs provide a very good outcome for most by decreasing the frequency of infections, clearing fluid and ventilating the space behind the eardrum,” he adds. “But some patients have issues with TTs becoming blocked. They can end up with a hole in the eardrum and scarring.”
Attending a presentation at a meeting of engineers, Remenschneider heard about the innovative materials science work at the Wyss Institute for Biologically Inspired Engineering at Harvard University. Wyss has focused on surface coatings to reduce algae and the adherence of molluscs to the boat hulls by impregnating them with a silicone oil. This provided the germ of an idea to impart non-stick properties to TTs.
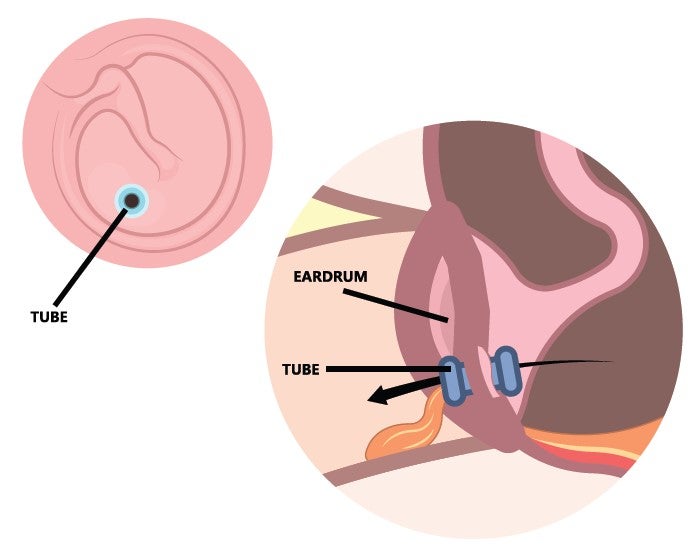
“Adding something like that to TTs would avoid some of the problems, so we explained what TTs are and the engineers immediately asked why they are the size and shape they are, why the flanges have a certain diameter and so on,” Remenschneider remarks. “I didn’t know. They have been designed that way for 50 years and have worked well enough, so nothing has changed, but the engineers immediately thought about geometry, coating, porosity and many other factors.”
The trouble with TTs
Though tympanostomy procedures are only performed in patients with chronic or treatment-resistant middle ear infections, the procedure is relatively common, so the major problems are well understood.
The function of plastic or metal TTs is known to be far from perfect, largely due to bacterial biofilms and local tissue growing on their surfaces. This blocks a TT’s lumen – the interior space through which fluids flow – and can cause them to extrude. This often occurs around one year after being implanted, though many are intended to stay in place far longer.
If squamous epithelial cells – a type of flat cell found throughout the body – accumulate beneath the tube’s outward flange, they can force the inward flange through the tympanic membrane into the external auditory canal. Sometimes, TTs fall out without notice, but even when they remain in place, they do not necessarily perform well.
One major limitation is that they are very poor at fluid flow. Fluid is sometimes unable to flow because of pinning, where fluid beads build up on the surface and needs high pressure to be driven through. If the flow is obstructed, antibiotic ear drops applied in the ear canal may not reach the site of infection. If frequent replacement surgeries are consequently required, costs rise significantly. Such problems are also common in other fluid-transporting implantable medical conduits (IMCs) – catheters, shunts and other small tubes used in the brain, liver and other organs. The challenge is that IMCs often need to be smaller in order to be used more effectively in less invasive procedures, but a decrease in size exacerbates problems with fluid flow.
“Being an ear doctor, I have thought about a lot of ear possibilities with the redesign of TTs, but here at MEE we also work on eyes, and there are IMCs that can work as shunts,” Remenschneider explains. “The existing technology has the same problems with fouling and occlusion.”
Through collaboration with the Wyss Institute, and the Harvard John A. Paulson School of Engineering and Applied Sciences (Seas), Remenschneider and the MEE team in Boston embarked on a project to completely overhaul IMC design. The result is a broadly applicable strategy that solves many of the problems that arise in making IMCs both smaller and more effective for uni and bi-directional fluid transport at the millimetre scale.
Total TT redesign
Remenschneider worked closely with Dr Joanna Aizenberg, an associate faculty member of the Wyss Institute, who has pioneered bioinspired materials including Slippery Liquid-Infused Porous Surfaces (Slips). With Slips, a thin layer of oil-based liquid is used to prevent biofouling.
Slips has already been used in many industrial applications, but Remenschneider and other physicians are now opening the door to its use in healthcare through a complete design overhaul of IMCs.
TTs manufactured with liquid-infused material – known as infused tympanostomy tubes (iTTs) – enable rapid drug delivery into the middle ear and improved fluid drainage. Furthermore, they limit water from crossing and entering from the outside, while also preventing bacterial and cell adhesion. This is partly due to a change in geometry, as the new design features a curved lumen.
Until now, all TTs have been designed with straight and cylindrical lumens, but the team hypothesised that specific curvatures could allow IMCs to discriminate between different fluids at a small scale.
“We took nothing for granted in the redesign,” Remenschneider says. “We went back to basics, and the engineers came up with many overly engineered solutions that were too complicated to manufacture or for clinicians to use, including designs with two lumens, but we arrived at a specific curved geometry and used an infused liquid material to pair with a particular fluid viscosity.”
The geometry decreases pinning, which happens mostly at sharp angles, and promotes fluid flow in one direction.
“The geometry is about fluid flow,” he adds. “Place it in one way to encourage flow out, and reverse it for flow in. The liquid infusion solves the problem of bacterial accretion. A biofilm that is hard to clear with antibiotics can sometimes build up, so we need to replace tubes when there is repeated infection, but with iTTs, cells have a hard time adhering to the tube, so a build up is less likely, and iTTs don’t work their way out.”
In six months of preclinical studies, no iTTs were extruded from the ear, suggesting they can dramatically reduce the need for tube replacement procedures.
“An ear tube is mainly just there to let air into the ear to clear fluid, but I think about other uses including drug delivery to the middle ear,” Remenschneider continues. “Some conditions need the direct application of steroids, which normally requires a syringe, anaesthetic and repeat visits. An implanted tube could allow patients to self-medicate.”
From iTTs to next-gen IMCs
The new design approach that created iTTs results in predictable and effective uni and bi-directional fluid transport at the millimetre scale and can resist contaminations, and could define how other IMCs are made. Indeed, the research team envisages the design methodology ultimately being applied to many different IMCs. At MEE, an aqueous shunt to decrease pressure in the eye is an obvious application, though the excellent safety and efficacy results in the ear lay the ground for shunts for the brain, bile duct, or for draining spinal fluid, and also suggest new designs for urinary catheters.
“We have been able to really define the materials and the performance in a preclinical model, so a human subject study is next in order to compare iTTs to standard-of-care tubes and assess the risk of biofilm formation over a longer duration of action,” Remenschneider explains.
“The materials are all commonly used and are known to be human-friendly, but any new device takes time, resources and data to reach the market,” he notes. “This could be transformative for our field, as preclinical data suggests that it could significantly reduce many of the complications we see with TTs. More exciting is that it could represent a new way to treat inner or middle ear conditions that have previously required invasive procedures.”
With new therapies developing fast, including gene therapies that could treat nerve-related hearing loss, the ability to get those medications into the inner ear is vital. Even without the potential applications elsewhere in the body, iTTs represent game-changing technology for millions of people.






